A Comparative Analytical and Invivo Evaluation of Musta (Cyperus Rotundus Linn.) and Shatavari (Asparagus Racemosus Wild.) for Anti Atherosclerotic Activity
DOI:
https://doi.org/10.47070/ayushdhara.v10i4.1001Keywords:
Musta, Shatavari, Antiatherosclerotic activity, New Zealand White rabbits.Abstract
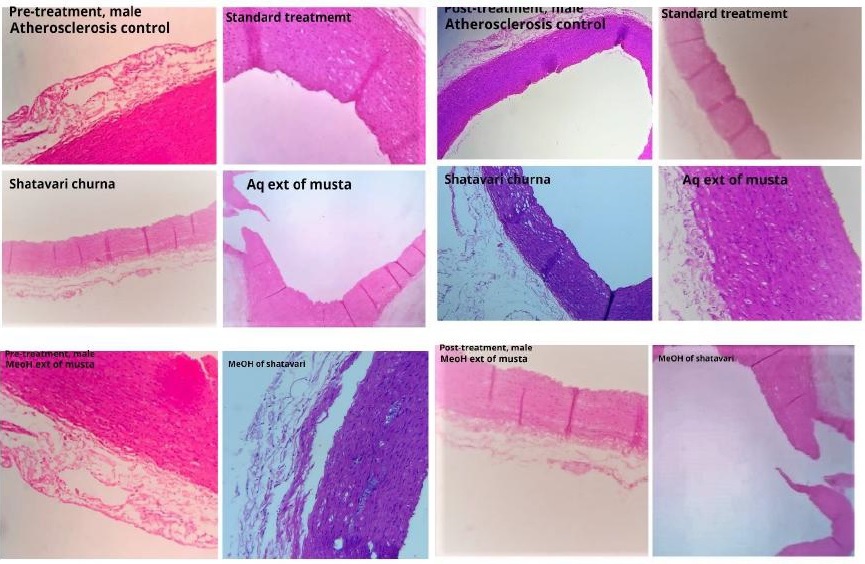
Obesity and hyperlipidemia are predisposing factors for major number of cardiovascular diseases, especially Atherosclerosis which has become the major cause for death. Sthoulya is a Medo pradoshaja vikara. Hyperlipidemia and Atherosclerosis can be correlated to Rasa pradoshaja vikara and Dhamani Prathichaya respectively. The treatment advocated for all the three conditions is Kaphahara and Medohara. Out of the big list of Kaphamedohara dravyas, Musta and Shatavari are two Dravyas with dissimilar Rasapanchakas that needs to be evaluated for the specific activity and indication. Analytical studies of the drugs were carried out as per the standard protocol. Invivo evaluation of Churna, Aqueous and Methanolic extracts of both the drugs in High fat diet induced Atherosclerosis were carried out on New Zealand White rabbits in both preventive and curative aspects. Though both the drugs were effective, Shatavari showed significant result in reducing Blood glucose, Total Cholesterol, Triglycerides, LDL and VLDL in comparison with Musta. Churna and Aqueous extract of Shatavari were effective than methanolic extract. Musta was found better in reducing the body weight in post treatment group than Shatavari. Churna and Methanolic extract of Musta were effective than aqueous extract in the reduction of body weight, Total cholesterol, Triglycerides, LDL and VLDL. Except body weight in pretreatment, the drugs were effective in both preventive and curative aspects. Musta is drug of choice in obesity associated with hyperlipidemia where as Shatavari would be effective in hyperlipidemia associated with Rasa kshaya lakshanas. Both Musta and Shatavari have potential Antiatherosclerotic activity.
Downloads
Downloads
Published
Issue
Section
License
Copyright (c) 2022 AYUSHDHARA

This work is licensed under a Creative Commons Attribution-NonCommercial-ShareAlike 4.0 International License.


